BHN English Toppage Company information OEM
Raw materials Support for overseas expansion

Company name: BHN Co., Ltd.
Head Office:1-16, Kanda Nishiki-cho, Chiyoda-ku, Tokyo, 101-0054, Japan
Harima M&D Center:1-472-41 Kohto Shingu-cho, Tatsuno, Hyogo, 679-5165, Japan
Yokohama Lab:75-1 Onocho Tsurumi-ku, Yokohama-shi, Kanagawa, 230-0046 Japan
- Established in Nov., 1999 (Sales launched on Jan., 2000)
- Capital: JPY80,000,000
- Director: CEO Mr. Akihiro Onda
Director: Mr. Yoichi Matsuura
Director: Mr. Takashi Kameyama
Director: Mr. Noriaki Fujii
Director: Mr. Atsushi Takii - Employee: 85 people * As of the end of August 2023
- Objective : Development, manufacturing, and sales of functional food ingredients, nutritional supplements, food additives, and cosmetics.
Company history
- Nov/1999
- Founded with JPY10,000,000 capital.
- Nov/1999
- Opened Osaka office.
- Jan/2000
- Starting sales activity
- Apr/2001
- Increased capital to JPY40,000,000.
- Jun/2001
- Started operation at Harima M&D Center.
- Aug/2003
- Increased capital to JPY60,000,000
- Sep/2003
- Certified with ISO9001
- Mar/2005
- Increased capital to JPY80,000,000
- Mar/2006
- Extended Harima M&D Center, and equipped new machine.
- Jul/2007
- Certified with health food GMP to packaging process.
- Jan/2013
- Started operation at Nagoya plant.
- Sep/2018
- Certifie with ISO22000
- Nov/2019
- BHN (Thailand) Co., Ltd. certified with ISO22000
- Sep/2021
- Concludes a stock transfer agreement and transfers management rights to Iwase Cosfa
- May/2023
- Opened Yokohama Lab
Group Company
BHN (Thailand) Co., Ltd. (Established in 2010, Thailand)
BHN Business
OEM
(Supplement,Cosmetics)
Raw Materials
(for Supplement, Food and Cosmetics)
Factory
Harima M&D Cenetr
BHN(Thailand) CO., Ltd
Research
Original Developments
Import materials
BHN supports customers’ request from ‘’market research’’, ‘’search for supply source’’, ‘’production’’ and
‘’own clinical trials’’. We hope we can make long-term relationship with business partners.
 |
Investigating market demand We will propose the best project to customers by consultation and market research. We will always try to be customer-oriented. |
 |
 |
Trial / Evaluation of project We will propose final product application and recipe by considering customers’ distribution and other products. We will confirm stability by appropriate test and establish shelf life. |
 |
 |
Safety and efficacy We will investigate all raw materials which will be used in final products. We will check legal compliance of all materialsincluding from bioactive substances to processing agent. We will also implement our own safety and efficacy test, if necessary. |
 |
 |
Production at network plant and QC We will select the best plant for production of final productsfrom our network,considering lot size, application and so on.Then, they will produce under our supervision. QC and final package of final products will be done in our Harima M&DCenter. |
 |
 |
Follow-up to make it ‘’strong product’’ We will not finish our projects by producing, but will contributeto make it strong products in the market. We will supply clinical trial evidence and advice marketing support. |
 |
Main Factory – Harima M&D Center

ISO 22000 ・Package line ・ Package line
2007: Healthy Food GMP (Package line)
2010: Permitted to produce cosmetics
2010: Certified as JAS organic
2018: Certified as ISO22000
BHN is producing final product packaging by strict QC measures



Unique technical development
BHN developed first multi-PTP line in Japan, and replies to customers’ various requests.
BHN (THAILAND) CO., LTD.
Capital : 55,000,000 Thai baht (about 1,700,000 USD)
Location : WHA Chonburi, Bo-win, Sriracha, Chonburi, Thailand
Business Lines : Production and distribution of Fish and Chicken Collagen,Supplement OEM


Policy
“3ST”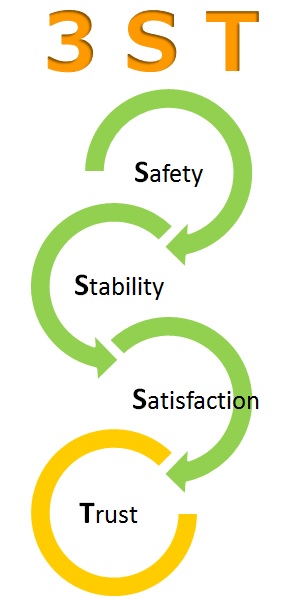
We are developing new products by R&D based on updated technical data and marketing concept.
Safety :
BHN don’t produce which is not proven to be safe. We investigate raw material in details and confirm safety.
Stability :
BHN confirms stability of products, and care about quality of final products for customers.
Satisfaction :
BHN produce the products with customer is satisfied under strict QC measure.
Trust :
Based on production complying with “Safety”, “Stability” and “Satisfaction”,BHN would like to become customers’ trust worthly business partner.
Company information OEM Raw materials Support for overseas expansion

